Menu
Physics Lesson 19.2.3 - Explanation of the Laws of Photoelectric Effect
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on Explanation of the Laws of Photoelectric Effect, this is the third lesson of our suite of physics lessons covering the topic of The Photoelectric Effect, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
Explanation of the Laws of Photoelectric Effect
From Einstein's Equation of Photoelectric Effect, it is obvious that this effect takes place only when photon's energy is greater than or equal to the work function (1st Law of Photoelectric Effect). Otherwise, electron could not be able to detach from metal surface. Therefore, the condition for the photoelectric effect to occur is that
or
The quantity Φ/f depends on the type of metal and it represents the threshold frequency for the photoelectric effect to occur. We denote this threshold frequency by f0, so we have:
We can calculate the value of threshold frequency for every metal shown in the table of work function provided in the previous paragraph. For example, the threshold frequency for iron (Φ = 4.36 eV,/sub>), is
= 4.36 ∙ 1.6 × 10-19 J/6.626× 10-34 J · s
= 1.05 × 1015 Hz
During the stoppage of photoelectrons by the electric field of stopping voltage, the work done by the electric forces is equal to the kinetic energy of the fastest photoelectrons, i.e.
where e is the elementary charge or the charge of electron (e = -1.6 × 10-19C) and ΔV0 is the stopping voltage. Thus, we have
Therefore, we obtain for the stopping voltage:
From the above formula, we see that there is a linear relationship between the stopping voltage and light frequency, i.e. the stopping voltage increases with the increase of light frequency (2nd Law of Photoelectric Effect). This relationship is shown in the graph below.
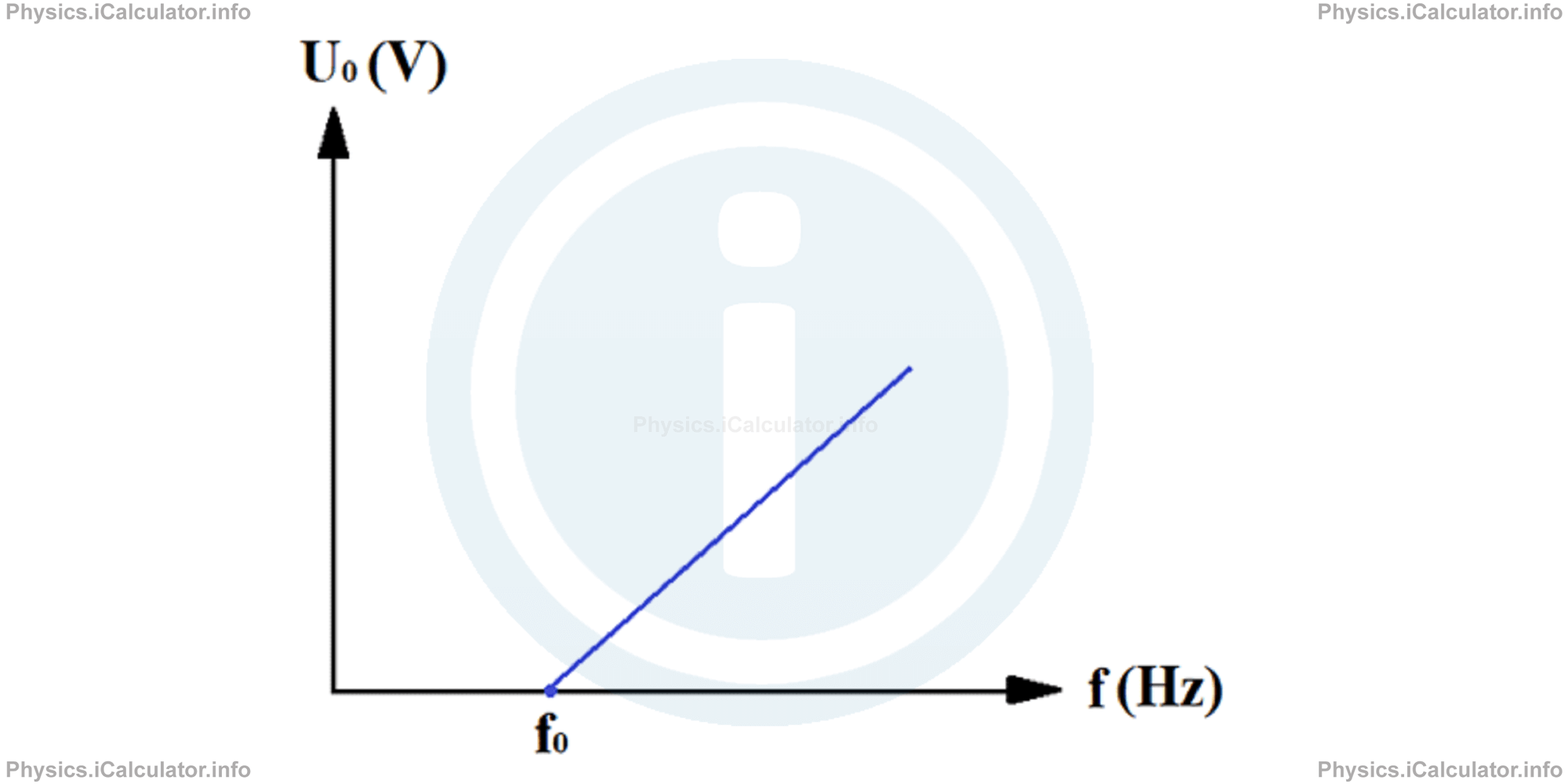
Photoelectric effect is a quantic phenomenon and as all the other quantic phenomena it is characterized by the probability of events occurrence. This means not all incident photons can cause photoelectric effect on a metal by detaching an electron from its surface. Some incident photons are absorbed by atoms of metals without producing any photoelectric effect while some other photons are reflected back by the metal surface. Thus, a photon can produce one or no photoelectrons when it falls on a metal.
If we denote by Nph the number of incident photons on the metal surface in one second and by Ne the number of photoelectrons produced in the same time, we have
This ratio (known as "quantic detachment caused by photoelectric effect") shows the probability for the photoelectric effect to occur. It is denoted by α and is a dimensionless quantity like all types of probability. Thus, we have
If α = 0, this means no electrons are detached from the surface of metal as the energy of all incident photons has been smaller than the work function.
If α = 1, this means each of the photons has detached one electron from metal. Theoretically, this occurs when all photons have a higher energy than work function. However, practically, this is impossible.
Photoelectrons that detach from cathode and reach the anode produce the photocurrent, which reaches the saturation value Is for specific values of accelerating voltage. The intensity of this saturation current is
= e ∙ α ∙ Nph
The number of photons incident on the cathode in every second is determined through the light flux Φ, and is given by
Hence, the saturation current is proportional to the light flux, as
This outcome represents the third law of photoelectric effect.
The fourth law states that photoelectric effect is a phenomenon without inertia. This is evident, based on the fact that the photon-electron interaction practically occurs at instant.
In this way, based on the quantum hypothesis on the particle nature of light, we managed to explain the laws of photoelectric effect found experimentally.
Example 2
What is the stopping voltage when a 500 nm photon falls on a sodium cathode (Φ = 2.26 eV)? Take the charge of electron e = 1.6 × 10-19 C.
Solution 2
Clues:
λ = 500 nm = 5 × 10-7 m
Φ = 2.26 eV = 2.26 × 1.9 × 10-19
J = 3.616 × 10-19 J
e = 1.6 × 10-19 C
(c = 3 × 108 m/s)
(h = 6.626 × 10-34 J · s)
First, we calculate the frequency of photon. Thus, giving that
we obtain for the frequency f of photon:
= 3 × 108 m/s/5 × 10-7 m
= 0.6 × 1015 Hz
= 6 × 1014 Hz
Now, using the equation
or
we obtain for the stopping voltage on the sodium cathode for the given frequency:
= 3.976 × 10-19J - 3.616 × 10-19 J/1.6 × 10-19 C
= 0.225 V
You have reached the end of Physics lesson 19.2.3 Explanation of the Laws of Photoelectric Effect. There are 3 lessons in this physics tutorial covering The Photoelectric Effect, you can access all the lessons from this tutorial below.
More The Photoelectric Effect Lessons and Learning Resources
Whats next?
Enjoy the "Explanation of the Laws of Photoelectric Effect" physics lesson? People who liked the "The Photoelectric Effect lesson found the following resources useful:
- Explanation Feedback. Helps other - Leave a rating for this explanation (see below)
- Modern Physics Physics tutorial: The Photoelectric Effect. Read the The Photoelectric Effect physics tutorial and build your physics knowledge of Modern Physics
- Modern Physics Revision Notes: The Photoelectric Effect. Print the notes so you can revise the key points covered in the physics tutorial for The Photoelectric Effect
- Modern Physics Practice Questions: The Photoelectric Effect. Test and improve your knowledge of The Photoelectric Effect with example questins and answers
- Check your calculations for Modern Physics questions with our excellent Modern Physics calculators which contain full equations and calculations clearly displayed line by line. See the Modern Physics Calculators by iCalculator™ below.
- Continuing learning modern physics - read our next physics tutorial: The Compton Effect and Pressure of Light
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "The Photoelectric Effect" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.
Modern Physics Calculators by iCalculator™
- Characteristic Em Wavelength Calculator
- De Broglie Wave Calculator
- Rayleigh Jeans Relation Calculator
- Energy Of Photons Calculator
- Intensity Photoelectric Effect Calculator
- Kinetic Photoelectric Effect Calculator
- Light Pressure Calculator
- Radiation Black Body Calculator
- Stopping Voltage Photoelectric Effect Calculator
- Uncertainty Calculator
- Wave Width Calculator
- Scattered Radiation Compton Effect Calculator
- De Broglie Packet Calculator