Menu
Physics Lesson 17.2.3 - Semiconductors
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on Semiconductors, this is the third lesson of our suite of physics lessons covering the topic of Electronic Components and Switching, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
Semiconductors
Electronic devices such as diodes and transistors are made from special materials called semiconductors. A semiconductor is a solid substance that has a conductivity between that of an insulator and that of most metals, either due to the addition of an impurity or because of temperature effects. Silicon (Si) and Germanium (Ge) [mainly Silicon] are the two most notorious examples of semiconductors used in electronics. Devices made of semiconductors, are essential components of most electronic circuits.
The reason why semiconductors are so widely used in electronics is the ability of such materials to host "impurities", which are extra electrons or extra "holes" (absence of electrons) in the semiconductors - a factor which contributes in the increase in their conducting ability. In other words, the addition of impurities to a semiconductor material leads to cause variation in the conducting nature of the material.
There are two types of impurities added to a semiconductor: donors and acceptors. The fundamental factor of difference between donor and acceptor impurities is that a donor impurity donates charges to the semiconductor while an acceptor impurity accepts the charges from the semiconductor material. The process of adding impurities to a semiconductor to increase its electricity conducting ability is known as "doping".
Semiconductors come in two primary types: intrinsic and extrinsic. Intrinsic semiconductors are semiconductors that are pure, i.e. they don't contain any doping agents added, whereas the extrinsic semiconductors do.
The adding of the doping agents changes the electron and hole carrier concentrations of the semiconductor at thermal equilibrium, i.e. at the temperature in which two adjacent substances do not exchange heat energy. Basically, it allows us manipulate the semiconductor to lower its resistance.
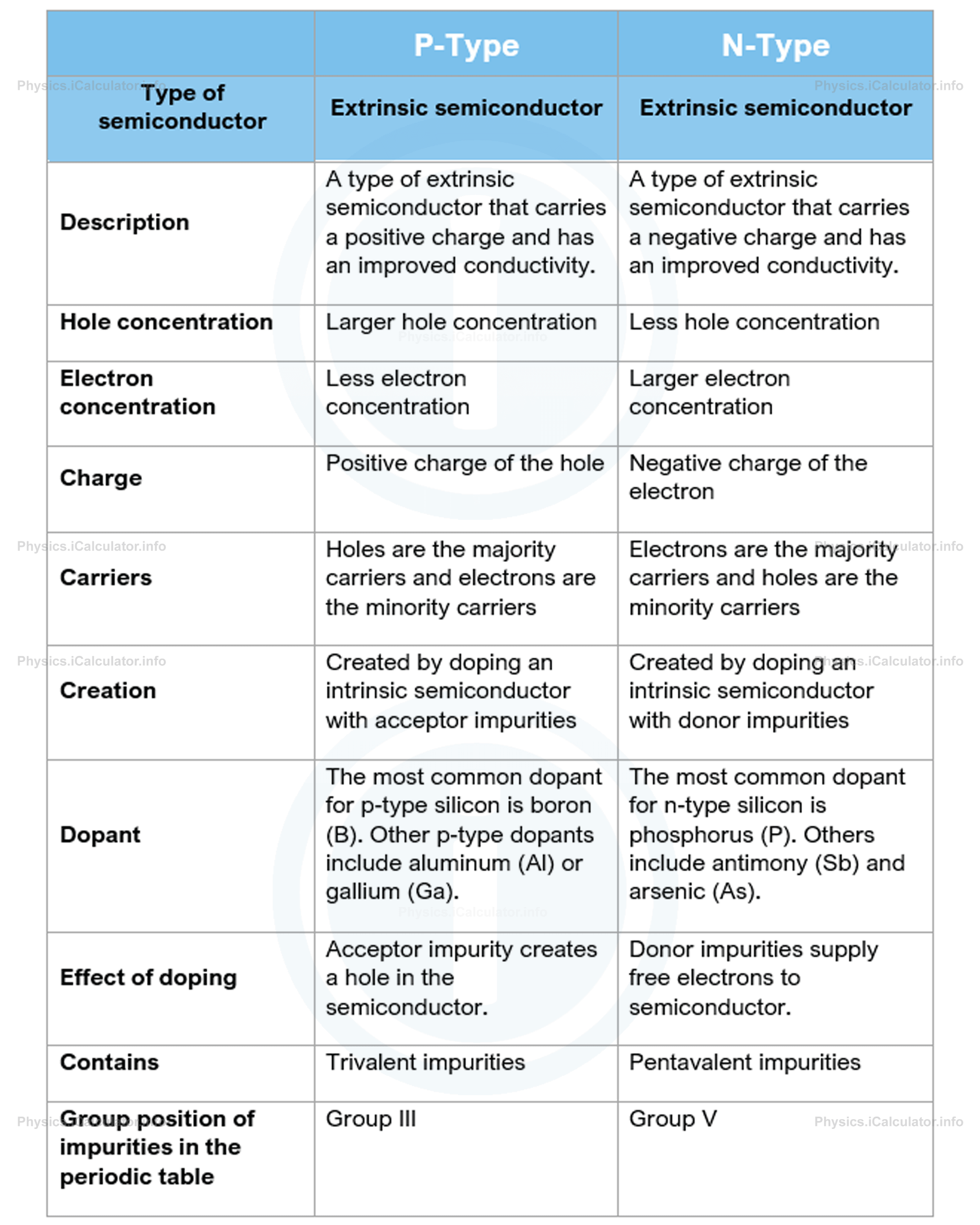
Extrinsic semiconductors have very common uses; they are mainly used as components in electrical devices where they provide high electrical resistance. Extrinsic semiconductors on the other hand, are essentially divided into two types: P-Type and N-Type. The P-Type semiconductors carry a positive charge, while the N-type ones carry a negative charge. This extra charge depends on the hole concentration and the electron concentration. Thus, a P-type semiconductor has a larger hole concentration, which results in a net positive charge. Similarly, a N-type semiconductor has a larger electron concentration than hole concentration, which results in a net negative charge.
Said this, it is obvious that a besides the main element, a P-type semiconductor contains a number of extra particles that belong to an acceptor-type impurity while a N-type semiconductor contains some extra particles that belong to a donor-type impurity.
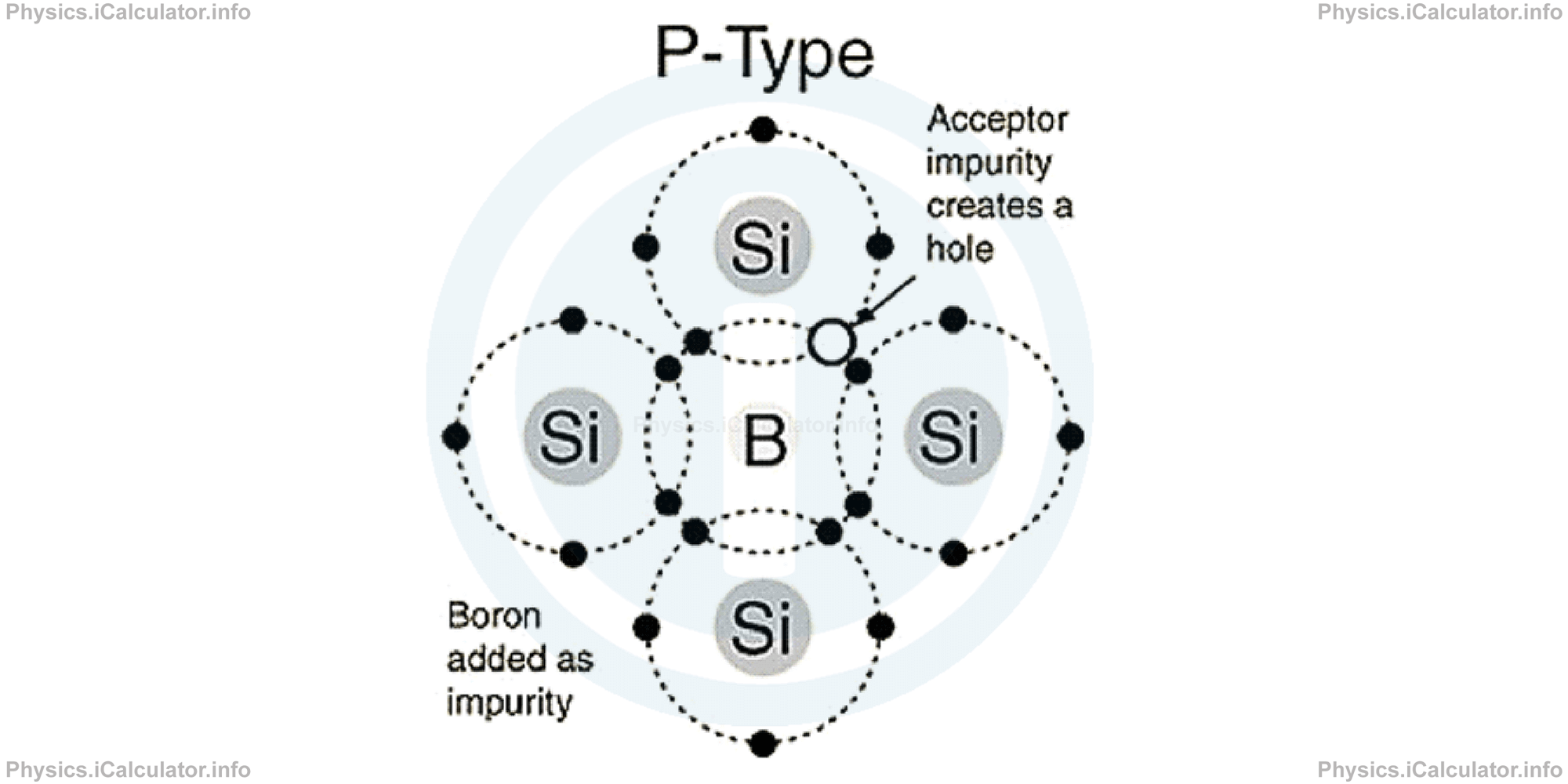
For example, when we add a Boron atom to Silicon, the Boron needs an extra electron to fill its outer layer because it is trivalent. It takes this extra electron from Silicon (or more exactly, a Silicon atom shares with the Boron atom an electron). In this way, a hole (lack of electron) is created in the material. This hole - which can shift throughout the material as this is a dynamic process - represents an extra positive charge created in the material, which acts as an electricity carrier, similarly to electrolyte, in which positive ions act as electricity carriers.
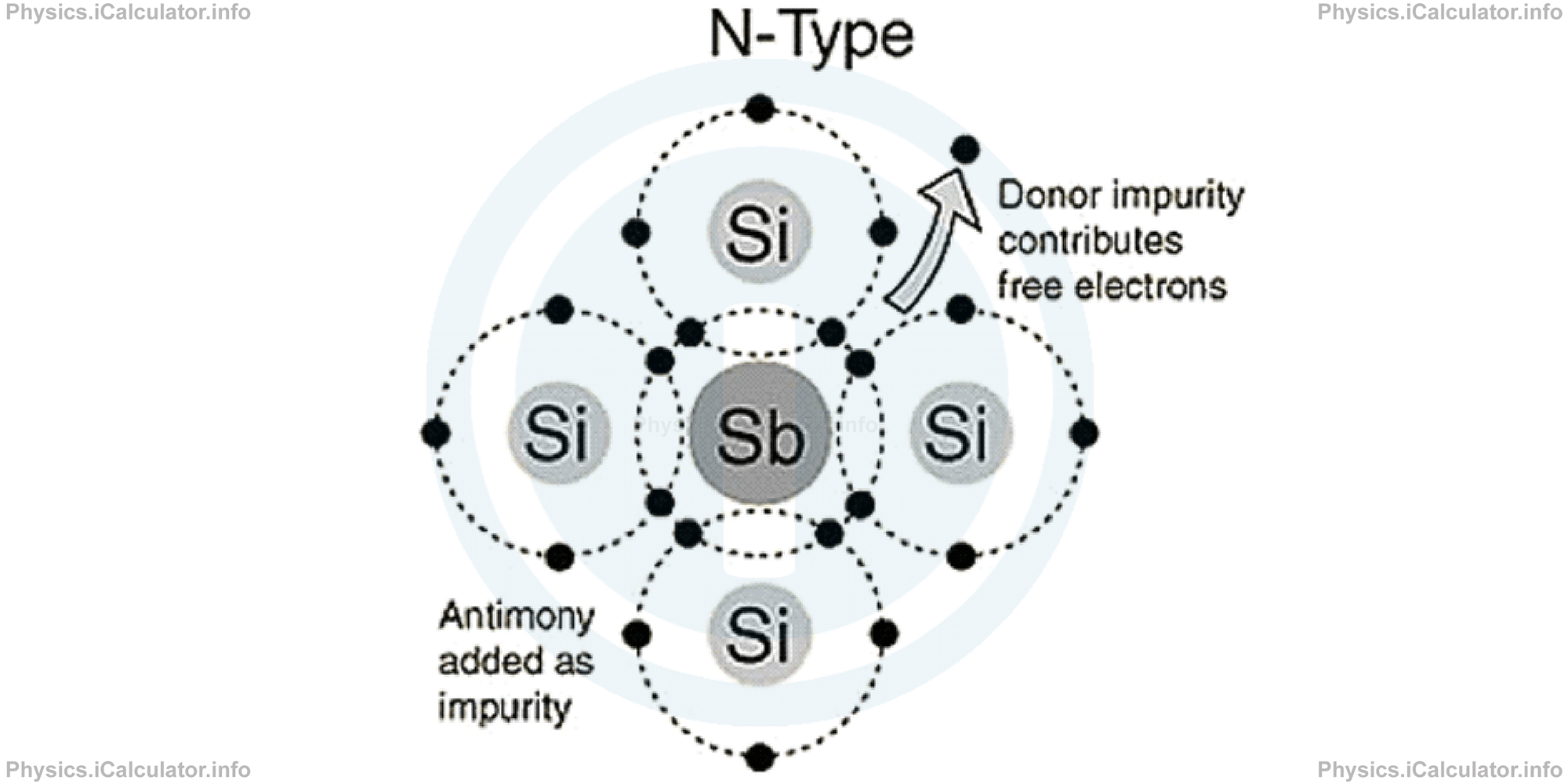
Likewise, when we add an Antimony (Sb) atom to Silicon, it brings an extra electron to the material (because it is pentavalent), which makes it negatively charged. This extra charge (electron) is a travelling charge, which displaces throughout the material.
The following table summarizes the main properties of semiconductors in regard to the extra charge they contain due to impurities.
Why are Semiconductors Preferred to Conductors in Electronics?
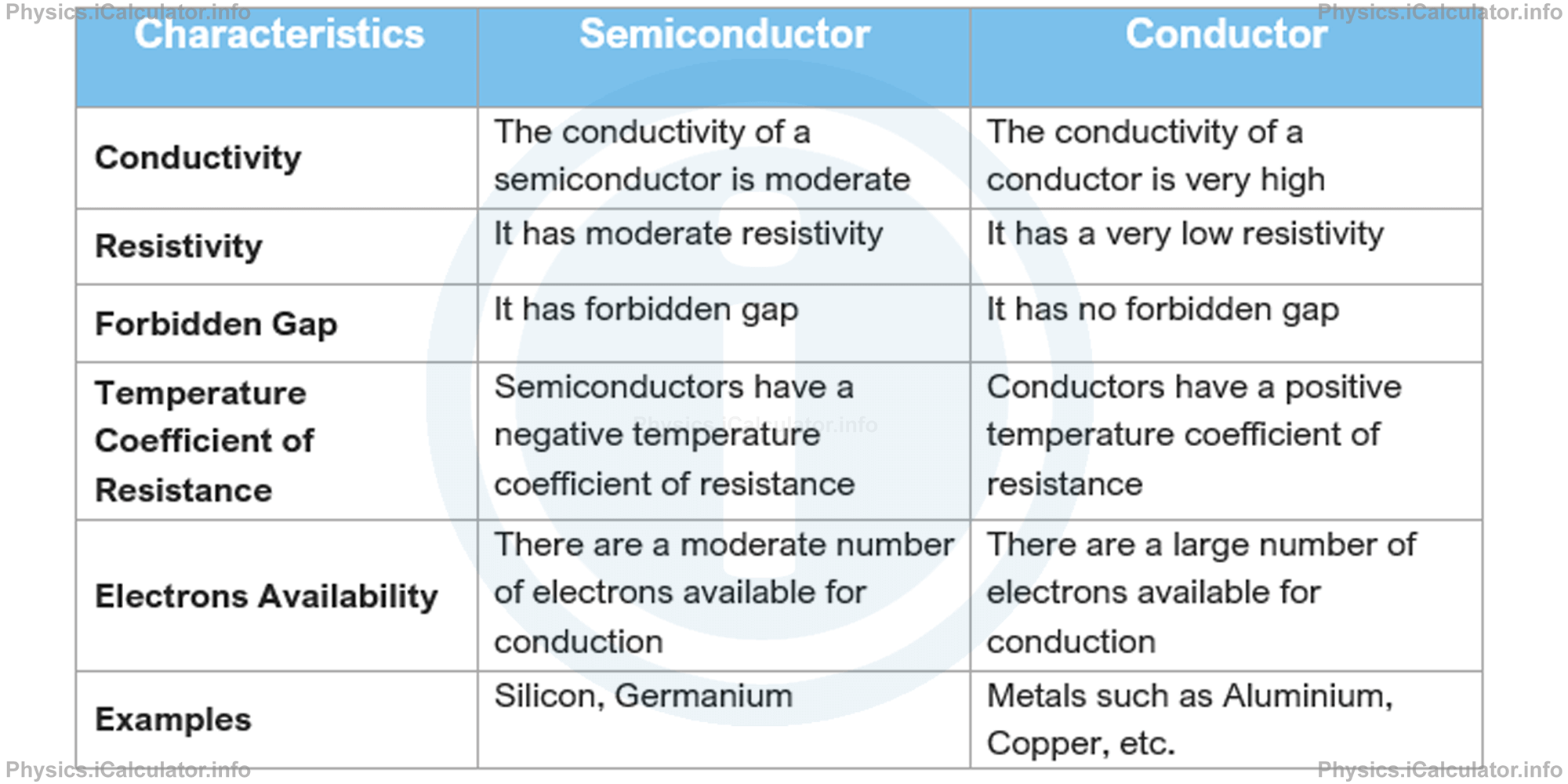
Semiconductors are more suitable than conductors in electronic circuits, as they need less power and can operate at very low voltage. Prior to introducing semiconductors in technology, mathematical operation and computation electronics were carried out by using "vacuum tube technology". Since the invention of transistors in 1947, semiconductors have played an important role in the electronic mathematical processing and computation.
Some of the advantages of semiconductors in electric circuit when compared to conductors include:
- Semiconductors need less area than conductors in an electronic circuit. In this way, they can fit in small devices such as mobile phones, tablets, etc.
- All electronics applications such as oscillator, amplification, mathematical operation can be executed through semiconductor devices such as diodes, transistor etc.
- Silicon element used in most electronic circuits is cheap and easily accessible.
- The capability of controlling the current flow by changing the voltage at another terminal is a huge benefit of semiconductor use in electronics.
- Semiconductor can acts both as a conductor and an insulator.
- In an extrinsic semiconductor, charge carriers increase in number not only by doping but also by the increase in temperature.
The difference between semiconductors and conductors is made clearer in the table below:
You have reached the end of Physics lesson 17.2.3 Semiconductors. There are 5 lessons in this physics tutorial covering Electronic Components and Switching, you can access all the lessons from this tutorial below.
More Electronic Components and Switching Lessons and Learning Resources
Whats next?
Enjoy the "Semiconductors" physics lesson? People who liked the "Electronic Components and Switching lesson found the following resources useful:
- Semiconductors Feedback. Helps other - Leave a rating for this semiconductors (see below)
- Electronics Physics tutorial: Electronic Components and Switching. Read the Electronic Components and Switching physics tutorial and build your physics knowledge of Electronics
- Electronics Revision Notes: Electronic Components and Switching. Print the notes so you can revise the key points covered in the physics tutorial for Electronic Components and Switching
- Electronics Practice Questions: Electronic Components and Switching. Test and improve your knowledge of Electronic Components and Switching with example questins and answers
- Check your calculations for Electronics questions with our excellent Electronics calculators which contain full equations and calculations clearly displayed line by line. See the Electronics Calculators by iCalculator™ below.
- Continuing learning electronics - read our next physics tutorial: Electronic Essentials: Analogue and Digital Signals, Binary Operations and Logic Gates
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "Electronic Components and Switching" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.