Menu
Physics Lesson 13.6.5 - Finding Work using a P - V Diagram
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on Finding Work using a P - V Diagram, this is the fifth lesson of our suite of physics lessons covering the topic of The Kinetic Theory of Gases. Ideal Gases, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
Finding Work from P - V Diagram
It is clear that in a P - V diagram, work is mathematically represented through the area under the graph. The simplest case is when pressure is constant as shown in the graph below.
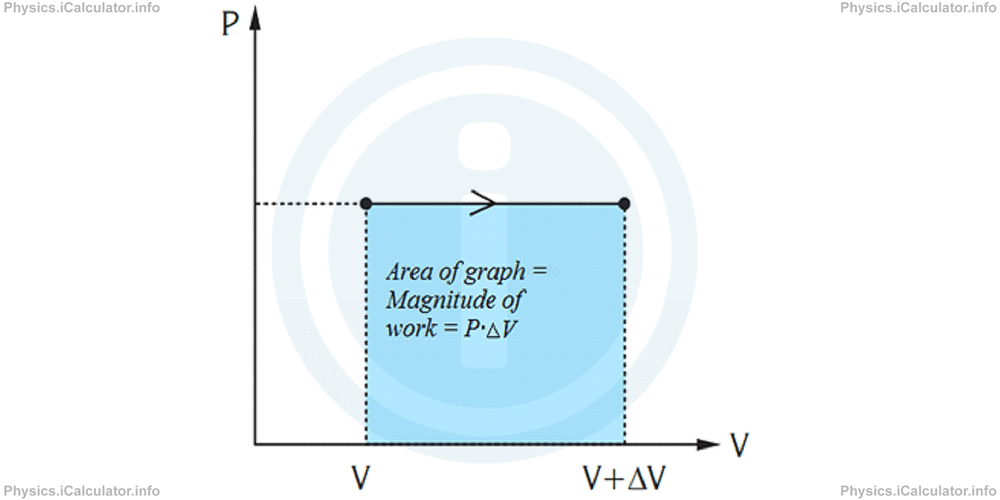
This reasoning can be extended in the processes with non-constant pressure as well. Thus, even if the pressure is changing, we can divide the curve over P - V diagram into small, constant pressure steps. The total area under the curve is therefore the sum of small rectangular areas. As we increase the number of rectangles by taking thinner rectangles, the error in calculation becomes smaller.
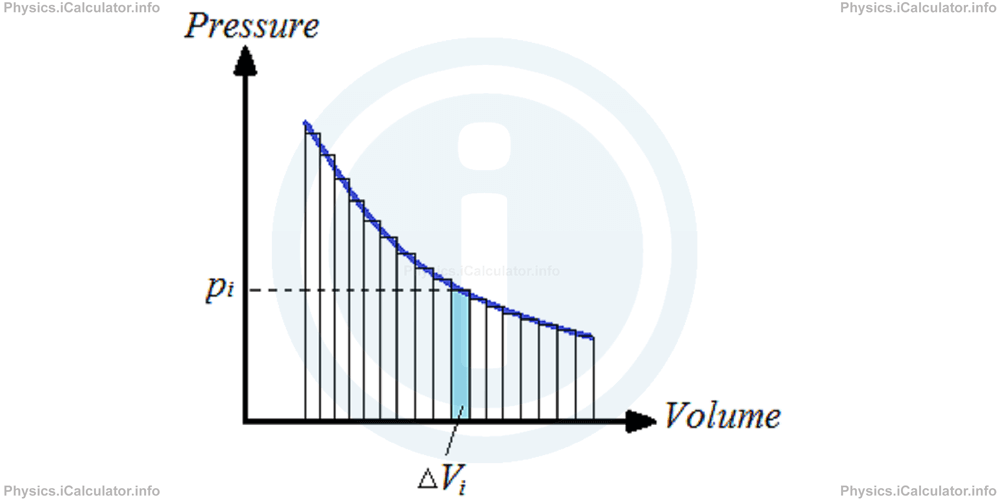
If we consider a certain rectangle (the i-th rectangle), the change in volume is ΔVi and the actual pressure is pi. Therefore, the work done to change the volume for this interval is
Thus, the sum of all such intervals gives the total work. If we divide the graph in n squares, we can write for the total work:
Example 5
A gas sample expands from 3 L to 11 L against the standard atmospheric pressure. What is the work done by the gas during this expansion?
Solution 5
Since the pressure is constant, we can use the P∆V formula to find the work done by the gas.
To find the result in joules, we must use the units Pa and m3. We know that the standard atmospheric pressure in pascals is Patm = 100 000 Pa. Also, we have V1 = 3 L = 0.003 m3 and V2 = 11 L = 0.011 m3. Therefore,
= 0.011 m3 - 0.003 m3
= 0.008 m3
Now, we can apply the work formula for constant volume:
= 100 000 Pa × 0,008 m3
= 800 J
You have reached the end of Physics lesson 13.6.5 Finding Work using a P - V Diagram. There are 6 lessons in this physics tutorial covering The Kinetic Theory of Gases. Ideal Gases, you can access all the lessons from this tutorial below.
More The Kinetic Theory of Gases. Ideal Gases Lessons and Learning Resources
Whats next?
Enjoy the "Finding Work using a P - V Diagram" physics lesson? People who liked the "The Kinetic Theory of Gases. Ideal Gases lesson found the following resources useful:
- P V Diagram Feedback. Helps other - Leave a rating for this p v diagram (see below)
- Thermodynamics Physics tutorial: The Kinetic Theory of Gases. Ideal Gases. Read the The Kinetic Theory of Gases. Ideal Gases physics tutorial and build your physics knowledge of Thermodynamics
- Thermodynamics Revision Notes: The Kinetic Theory of Gases. Ideal Gases. Print the notes so you can revise the key points covered in the physics tutorial for The Kinetic Theory of Gases. Ideal Gases
- Thermodynamics Practice Questions: The Kinetic Theory of Gases. Ideal Gases. Test and improve your knowledge of The Kinetic Theory of Gases. Ideal Gases with example questins and answers
- Check your calculations for Thermodynamics questions with our excellent Thermodynamics calculators which contain full equations and calculations clearly displayed line by line. See the Thermodynamics Calculators by iCalculator™ below.
- Continuing learning thermodynamics - read our next physics tutorial: Pressure, Temperature and RMS Speed
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "The Kinetic Theory of Gases. Ideal Gases" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.
Thermodynamics Calculators by iCalculator™
- Carnot Engine Efficiency Calculator
- Entropy Calculator
- Gas Laws Calculator
- Molecular Mean Free Path Calculator
- Translational Kinetic Energy Of Gas Calculator
- Root Mean Square Speed Calculator
- Ideal Gas Law Calculator
- Change In The Gas Internal Energy Calculator
- Radiative Heat Transfer Calculator
- Evaporative Heat Transfer Calculator
- Convective Heat Transfer Calculator
- Conductive Heat Transfer Calculator
- Final Temperature Of Mixture Calculator
- Heat Absorbed Or Released Calculator
- Thermal Expansion Calculator
- Temperature Calculator