Menu
Physics Lesson 20.1.2 - Thompson's Atomic Model (in 1898)
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on Thompson's Atomic Model (in 1898), this is the second lesson of our suite of physics lessons covering the topic of Atomic Nucleus and Its Structural Properties, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
Thompson's Atomic Model (in 1898)
According to the Thomson atomic model, often referred to as the "plum-pudding" model, the atom is a sphere of uniformly distributed positive charge about one angstrom in diameter (1 angstrom = 0.1 nm = 10-10 m). Electrons are embedded in a regular pattern, like raisins in a plum pudding or watermelon, to neutralize the positive charge.
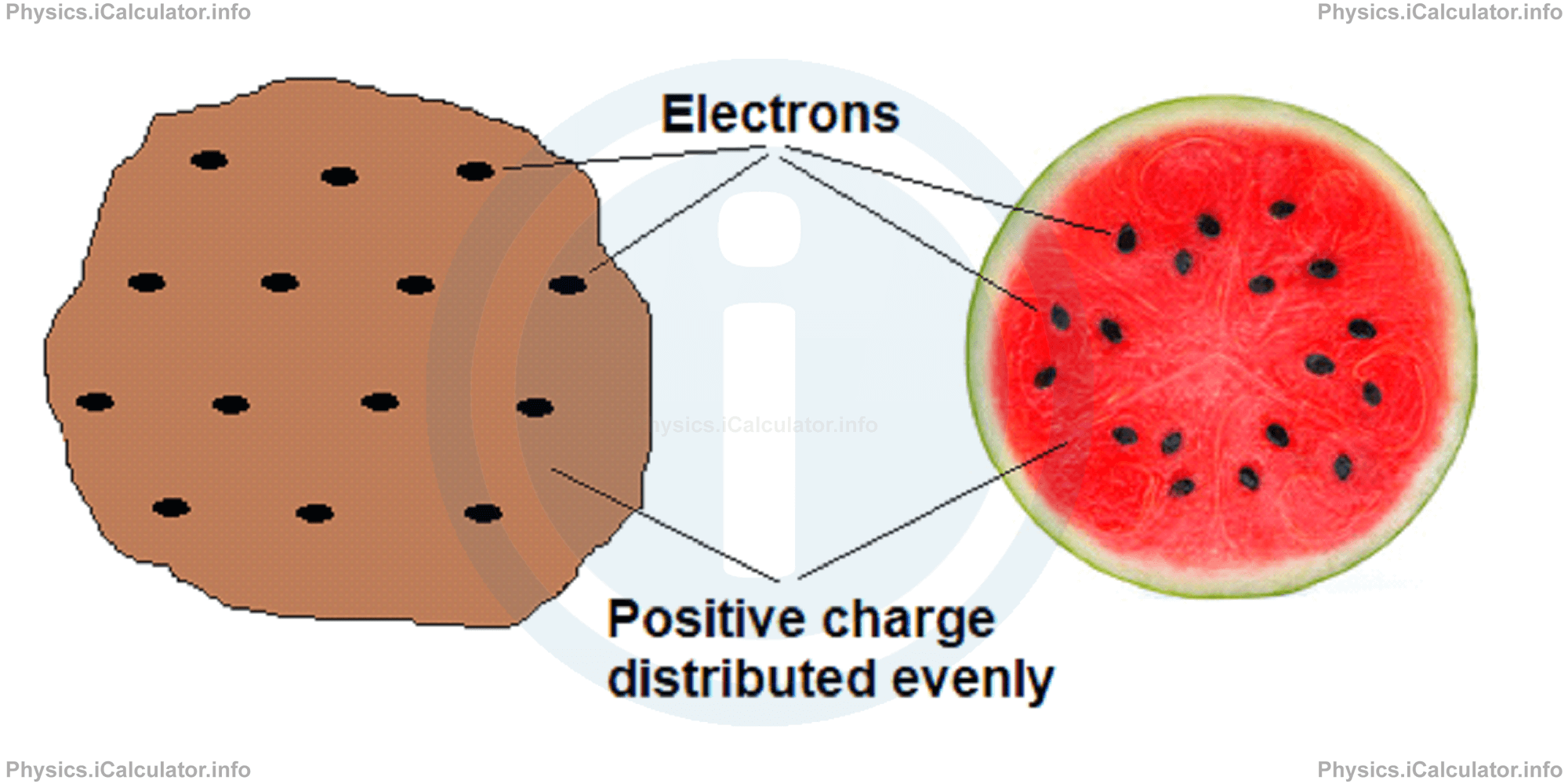
However, now we know that this model is wrong, as the true atomic structure is completely different. The key discovery in this regard was made in 1911, with the experiment carried out by Rutherford, which we will explain in the next paragraph.
You have reached the end of Physics lesson 20.1.2 Thompson's Atomic Model (in 1898). There are 9 lessons in this physics tutorial covering Atomic Nucleus and Its Structural Properties, you can access all the lessons from this tutorial below.
More Atomic Nucleus and Its Structural Properties Lessons and Learning Resources
Whats next?
Enjoy the "Thompson's Atomic Model (in 1898)" physics lesson? People who liked the "Atomic Nucleus and Its Structural Properties lesson found the following resources useful:
- Thompson Feedback. Helps other - Leave a rating for this thompson (see below)
- Nuclear Physics Physics tutorial: Atomic Nucleus and Its Structural Properties. Read the Atomic Nucleus and Its Structural Properties physics tutorial and build your physics knowledge of Nuclear Physics
- Nuclear Physics Revision Notes: Atomic Nucleus and Its Structural Properties. Print the notes so you can revise the key points covered in the physics tutorial for Atomic Nucleus and Its Structural Properties
- Nuclear Physics Practice Questions: Atomic Nucleus and Its Structural Properties. Test and improve your knowledge of Atomic Nucleus and Its Structural Properties with example questins and answers
- Check your calculations for Nuclear Physics questions with our excellent Nuclear Physics calculators which contain full equations and calculations clearly displayed line by line. See the Nuclear Physics Calculators by iCalculator™ below.
- Continuing learning nuclear physics - read our next physics tutorial: Nuclear Forces, Defect of Mass and Binding Energy
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "Atomic Nucleus and Its Structural Properties" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.