Menu
Physics Lesson 15.4.2 - Sources of Electricity
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on Sources of Electricity, this is the second lesson of our suite of physics lessons covering the topic of Electric Circuits. Series and Parallel Circuits. Short Circuits, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
Sources of Electricity
As stated earlier, in order to keep a steady flow of electrons through the circuit, we need a device that pushes the electrons and makes them flow continuously through the circuit. There are two major groups of devices, which can achieve this goal. The first group consists on batteries that produce one-directional current or direct current, (DC) and the other group consists on electric generators that produce alternating current (AC), i.e. a kind of current which charges its direction continuously (60 times per second in most countries of Americas and 50 times per second in the rest of the world). Here, we will focus on the DC current because AC current will be discussed in the next chapter.
As explained earlier, a cell is the basic element of a battery. Therefore, in this article we will explain the operating principle of various types of cells only. Basically, a cell converts chemical energy into electricity. There are three main types of cells:
a. Wet (voltaic) cells
A wet cell is made up by a copper and a zinc plate immersed inside a weak acid solution. The acid solution is called electrolyte and it is used to conduct electricity, while the plates are called electrodes. The free electrons emerge from the zinc plate (anode) and move to the copper plate (cathode) when a conducting wire connects the two electrodes. As a result, an electric current is produced.
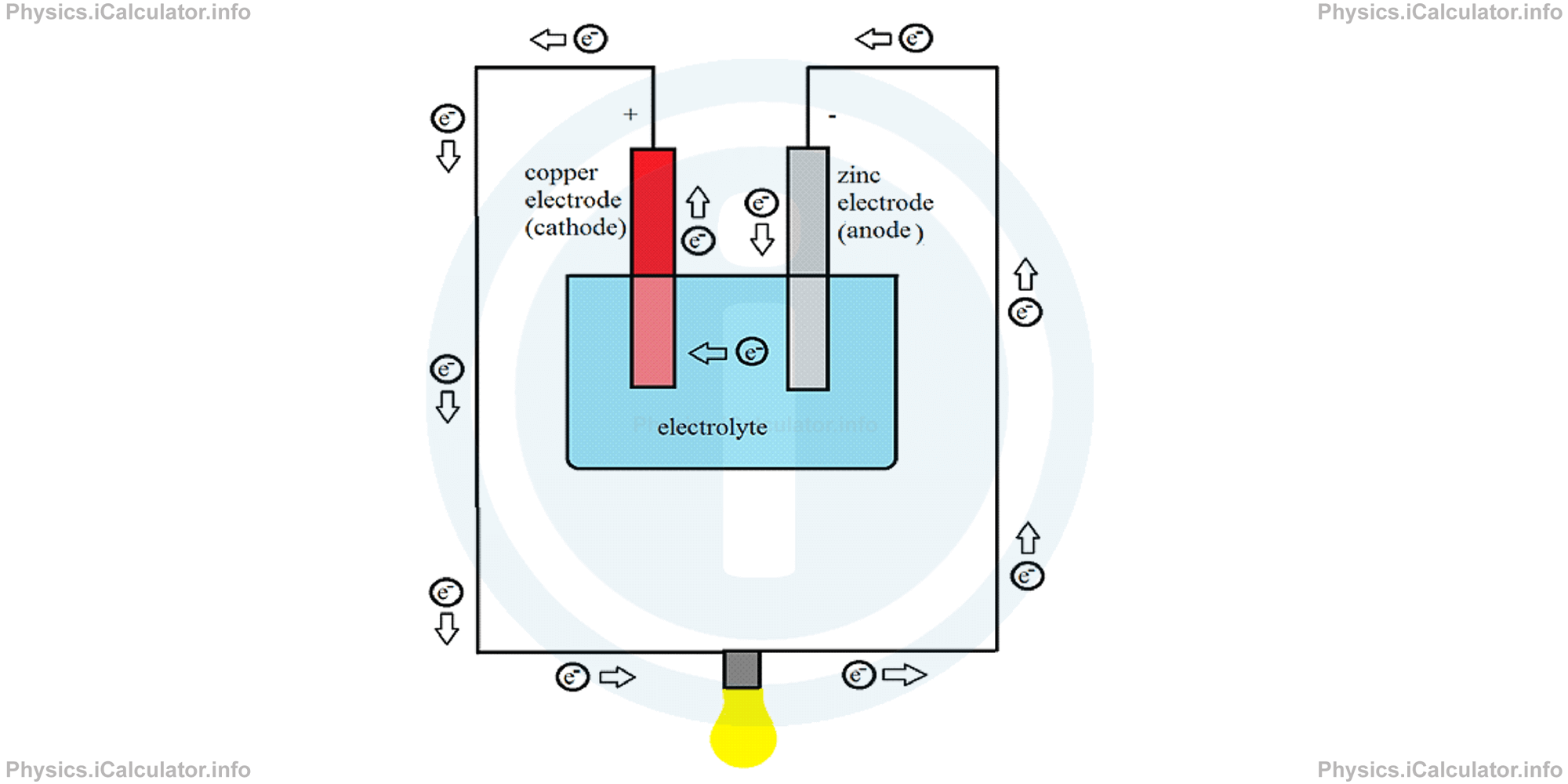
This type of cell can produce a 1V potential difference. Here, the carriers of electricity inside the electrolyte are positive and negative ions while electrons are carriers of electricity in the rest of the circuit. The electrolyte "scours" the electrodes and makes positive ions move from zinc to copper electrode while negative ions move in the opposite direction. As a result, the zinc plate is charged positively and copper plate negatively. The flow of electrons through the conducting wire tends to set up the balance by transferring electrons from zinc to copper. However, the scouring effect of electrolyte meanwhile continues, resulting in new ions produced. This process lasts for a long time, producing as a result a steady current in the circuit.
Wet cells are the oldest type of cells. The first wet cell was invented by Alessandro Volta, an Italian scientist who used salt-soaked leather as an electrolyte. Therefore, the unit of potential difference bears his name (Volt) in honour to his work.
b. Dry cells
Wet cells have many limitations, from the low potential difference they produce (1V) to the difficulty in transportation due to the acidic liquid contained in them. Therefore, during their attempts to improve the battery, scientists invented another type of cell - the dry cell.
A dry cell consists of a zinc cylindrical can (anode) and a carbon rod at the centre (cathode). A chemical paste here substitutes the effect of acidic solution used in the wet cells.
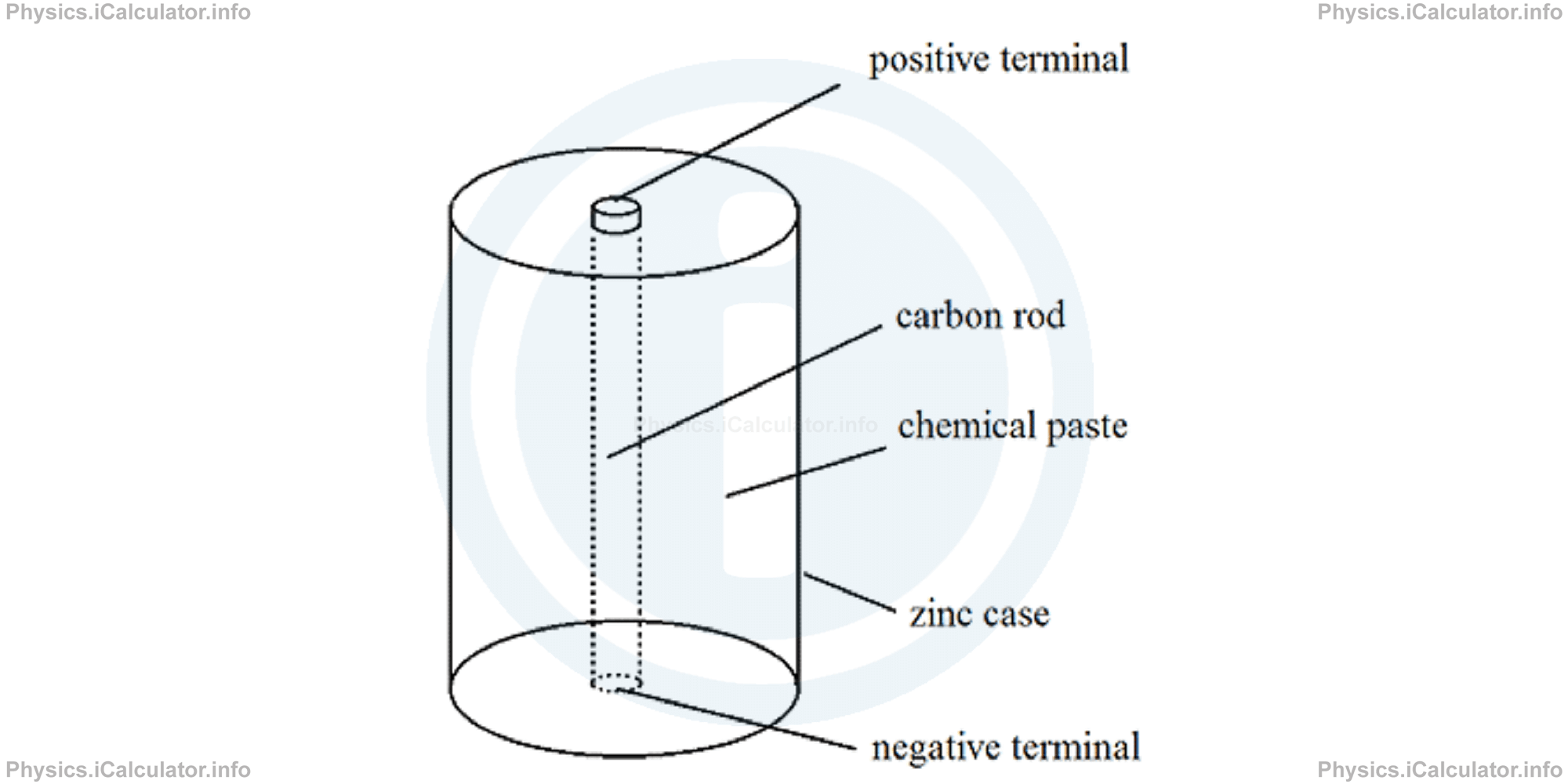
The operating principle of dry cells is the same as in wet cells. Dry cells produce a potential difference of 1.5 V (50% more than wet cells); furthermore, they are easy to transport as you can rotate them without problem.
c. Accumulators
Accumulators are also known as "storage batteries." They are used in cars, laptops, flashlights etc. Accumulators are groups of wet cells enclosed inside plastic containers. Zinc and copper electrodes here are replaced by lead electrodes. Each cell of accumulators produce 2V of potential difference. This is the reason why they are preferable when a higher voltage is needed.
There are 6 cells accumulators (in total they produce 12 V) and 9 cells accumulators (in total they produce 18 V). In addition, the electrolyte of accumulators is made up from dilute sulphuric acidic solution.
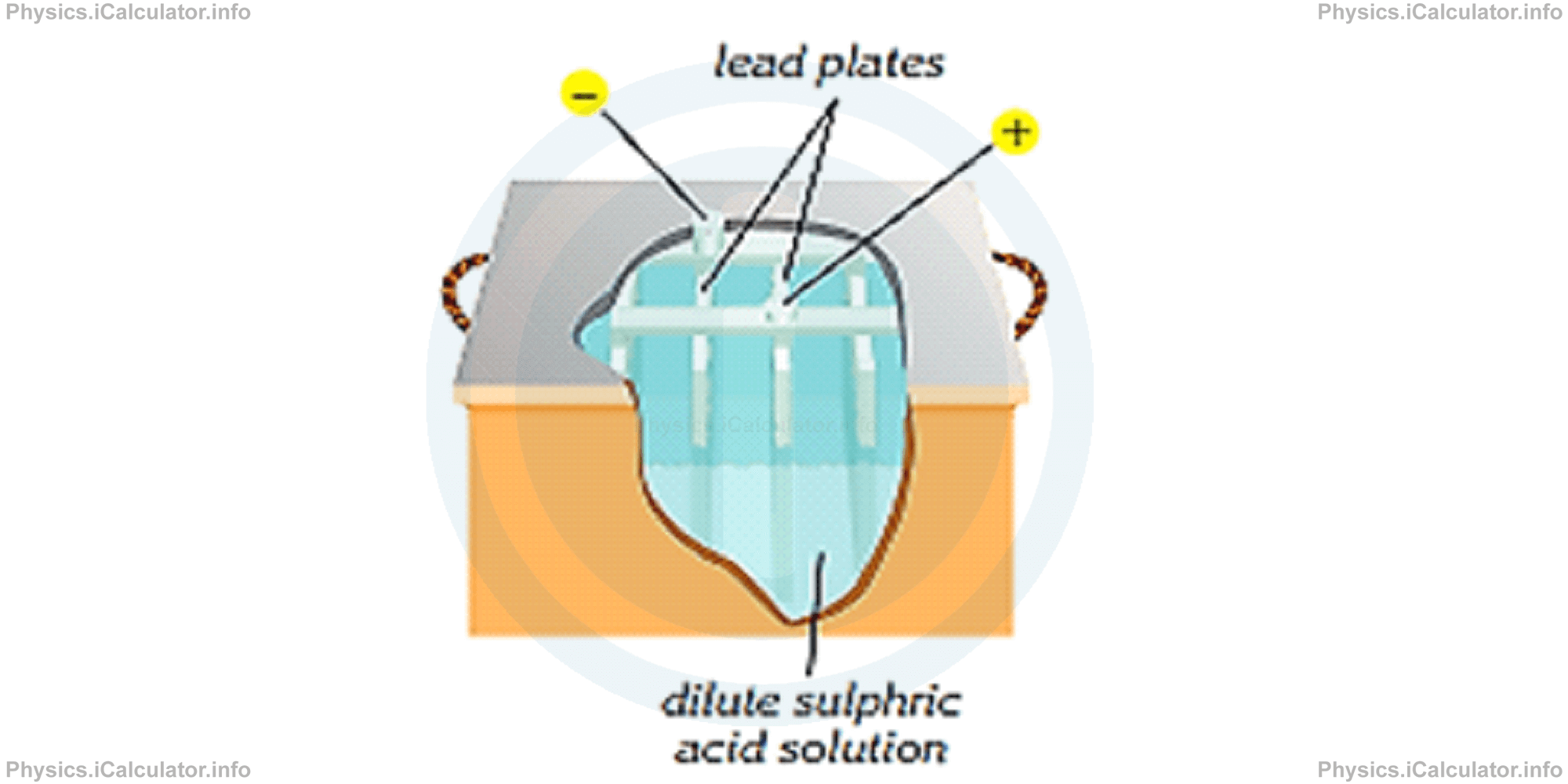
Accumulators have an important advantage: they are all rechargeable.
You have reached the end of Physics lesson 15.4.2 Sources of Electricity. There are 5 lessons in this physics tutorial covering Electric Circuits. Series and Parallel Circuits. Short Circuits, you can access all the lessons from this tutorial below.
More Electric Circuits. Series and Parallel Circuits. Short Circuits Lessons and Learning Resources
Whats next?
Enjoy the "Sources of Electricity" physics lesson? People who liked the "Electric Circuits. Series and Parallel Circuits. Short Circuits lesson found the following resources useful:
- Sources Feedback. Helps other - Leave a rating for this sources (see below)
- Electrodynamics Physics tutorial: Electric Circuits. Series and Parallel Circuits. Short Circuits. Read the Electric Circuits. Series and Parallel Circuits. Short Circuits physics tutorial and build your physics knowledge of Electrodynamics
- Electrodynamics Revision Notes: Electric Circuits. Series and Parallel Circuits. Short Circuits. Print the notes so you can revise the key points covered in the physics tutorial for Electric Circuits. Series and Parallel Circuits. Short Circuits
- Electrodynamics Practice Questions: Electric Circuits. Series and Parallel Circuits. Short Circuits. Test and improve your knowledge of Electric Circuits. Series and Parallel Circuits. Short Circuits with example questins and answers
- Check your calculations for Electrodynamics questions with our excellent Electrodynamics calculators which contain full equations and calculations clearly displayed line by line. See the Electrodynamics Calculators by iCalculator™ below.
- Continuing learning electrodynamics - read our next physics tutorial: Kirchhoff Laws
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "Electric Circuits. Series and Parallel Circuits. Short Circuits" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.
Electrodynamics Calculators by iCalculator™
- Amount Of Substance Obtained Through Electrolysis Calculator
- Charge Density Calculator
- Electric Charge Stored In A Rc Circuit Calculator
- Electric Field In Terms Of Gauss Law Calculator
- Electric Power And Efficiency Calculator
- Electron Drift Velocity Calculator
- Equivalent Resistance Calculator
- Force Produced By An Electric Source Calculator
- Joules Law Calculator
- Ohms Law Calculator
- Potential Difference In Rc Circuit Calculator
- Resistance Due To Temperature Calculator
- Resistance Of A Conducting Wire Calculator