Menu
Physics Lesson 9.1.1 - The Definition of Matter. States of Matter and their Features
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on The Definition of Matter. States of Matter and their Features, this is the first lesson of our suite of physics lessons covering the topic of Fluids. Density of Fluids, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
The Definition of Matter. States of Matter and their Features
Matter is described as a "physical substance in general, which occupies space and possesses rest mass, especially as distinct from energy."
In simpler words, matter is composed by the combination of a very large number of small particles known as "atoms". In a certain sense, atoms are the "building blocks" of matter. The dimensions of atoms vary from 0.1 to 0.5 nm (1 nm = 10-9 m). To give an idea how small atoms are, we can mention the fact that an atom is about one million times smaller than the average thickness of human hair.
Matter can exist in three stable states in nature (in fact, there are five states of matter but three are common to us). They are:
1- Solid state
In this state, particles are "packed" in stable positions, which they cannot leave. In other words, particles in solid state cannot change their neighbours. They always have the same particles around them.
If we use an energetic approach to explain the states of matter, we must stress that in solid state the potential energy that keeps the particles together, which is also known as the "binding energy" is much greater than kinetic energy of particles. As a result, atoms in solids can only vibrate around their equilibrium position but they cannot leave their actual place and find new neighbouring atoms.
The most suitable pattern used to describe the solid state is a cube in whose vertexes some spheres are placed and whose sides are made of elastic springs as shown in the figure below.
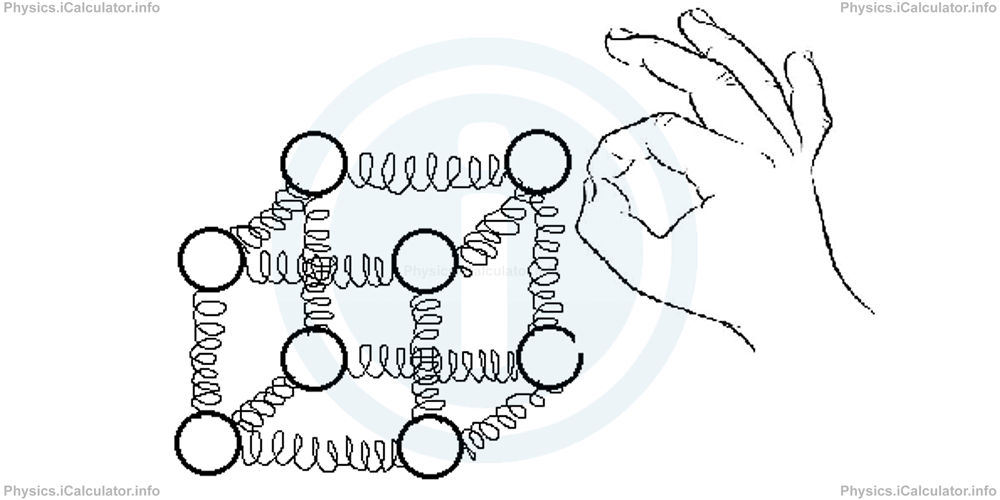
If you kick one of spheres with your finger, energy is transferred from the hand to the first sphere and then to all the other spheres through the elastic springs. As a result, all spheres will start vibrating around their equilibrium position. This is more or less what happens with particles of solids in the micro-world.
2- Liquid state
In this state, particles are still very close but they are not "attached" together. Their potential energy is still a bit greater than kinetic energy but the difference between them is very narrow. As a result, particles can slide easily over each other, like plastic balls when poured on a playground. Look at the figure.
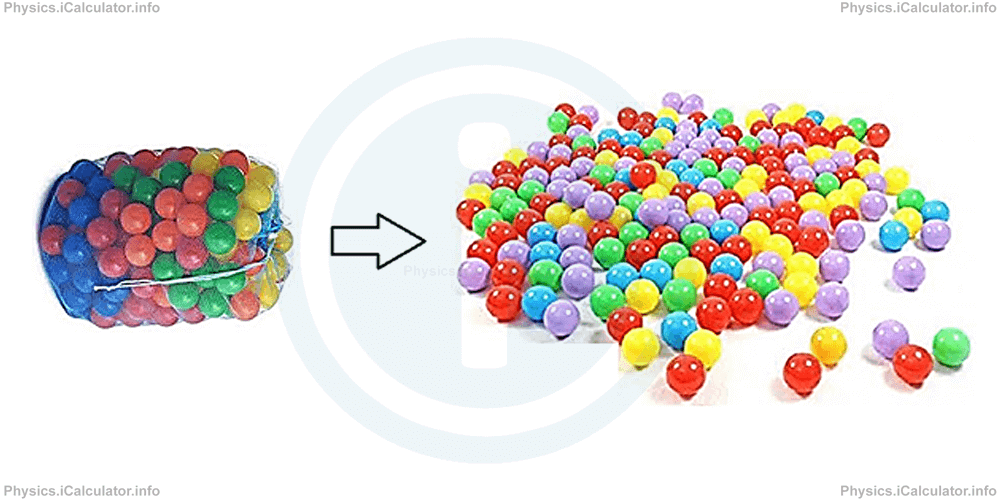
The balls distribute throughout the lower base of container due to gravity. They cannot accumulate on top of each other until the first layer is completely filled with balls. Then, the second layer start to form and so on. The same thing occurs in liquids as well. This is the reason why the surface of a liquid is flat.
Gaseous state
In this state, particles move freely in space as their kinetic energy is greater than the potential (binding) energy. They look like balloons flying in the sky or like bubbles flowing in water. Look at the figure.

You have reached the end of Physics lesson 9.1.1 The Definition of Matter. States of Matter and their Features. There are 5 lessons in this physics tutorial covering Fluids. Density of Fluids, you can access all the lessons from this tutorial below.
More Fluids. Density of Fluids Lessons and Learning Resources
Whats next?
Enjoy the "The Definition of Matter. States of Matter and their Features" physics lesson? People who liked the "Fluids. Density of Fluids lesson found the following resources useful:
- Definition Feedback. Helps other - Leave a rating for this definition (see below)
- Density and Pressure Physics tutorial: Fluids. Density of Fluids. Read the Fluids. Density of Fluids physics tutorial and build your physics knowledge of Density and Pressure
- Density and Pressure Revision Notes: Fluids. Density of Fluids. Print the notes so you can revise the key points covered in the physics tutorial for Fluids. Density of Fluids
- Density and Pressure Practice Questions: Fluids. Density of Fluids. Test and improve your knowledge of Fluids. Density of Fluids with example questins and answers
- Check your calculations for Density and Pressure questions with our excellent Density and Pressure calculators which contain full equations and calculations clearly displayed line by line. See the Density and Pressure Calculators by iCalculator™ below.
- Continuing learning density and pressure - read our next physics tutorial: Pressure. Solid Pressure
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "Fluids. Density of Fluids" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.