Menu
Physics Lesson 9.7.1 - Adhesive and Cohesive Forces
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on Adhesive and Cohesive Forces, this is the first lesson of our suite of physics lessons covering the topic of Adhesive and Cohesive Forces. Surface Tension and Capillarity, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
Adhesive and Cohesive Forces
As explained in Physics tutorial "Density. Density of Fluids", matter is made up by microscopic particles called atoms. Sometimes, two or more atoms pair together in molecules. In solids, particles are strongly bonded to each other, while in gases their bonding is very weak. Liquids are an intermediate category between solids and gases.
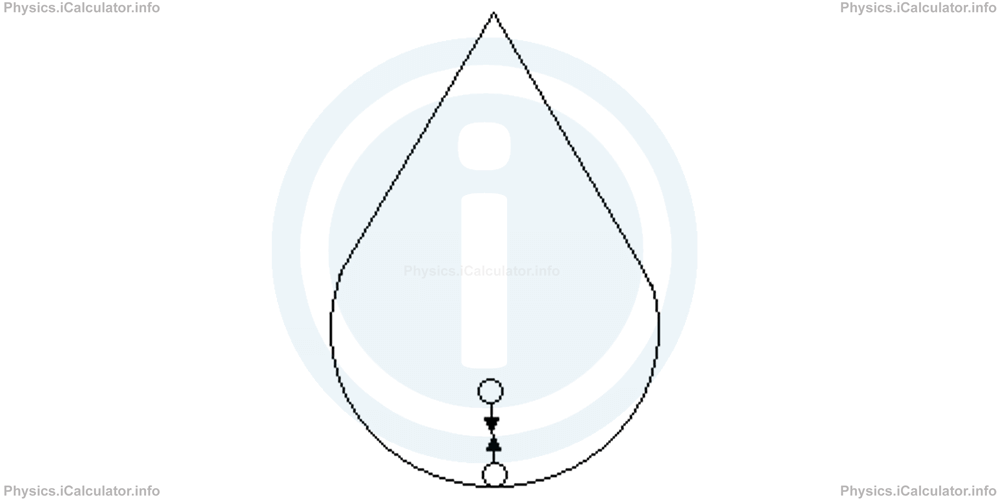
By definition, cohesion is the property of like particles to stick to each other. Obviously, these like particles belong to the same substance. To make possible such a mutual attraction, particles interact with each other through certain attraction forces known as cohesive forces. For example, water droplets take a spherical shape in the lower part as cohesive forces exerted by inner molecules to those in the surface, cause a shrink on water volume and as a result, all water molecules tend to gather around the centre of the droplet.
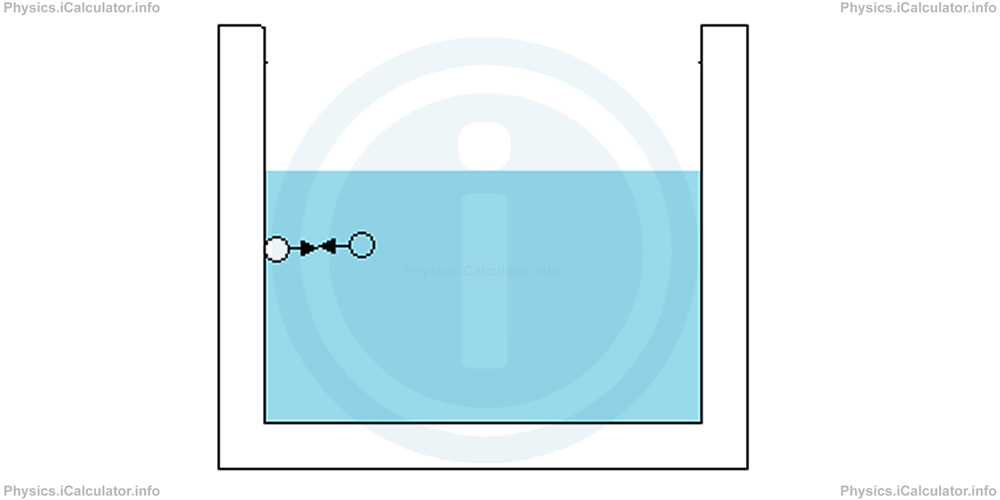
On the other hand, when substances are different, they stick to each other because of adhesive forces. Thus, we can define adhesion as the ability of different substances or surfaces to cling to each other. For example, water wets the inner part of a metal container because water molecules cling to the molecules of the metal container by means of adhesive forces. Another example: water bubbles gather to the inner faces of the container because of adhesive forces of water.
The existence of adhesive and cohesive forces depends in great extent by the densities of surfaces in contact. Thus, in solids (which usually have high densities), cohesive forces prevail over adhesive ones as molecules of solids are closer (and as a result, stronger bonded) to each other than to the molecules of the other substance they are in contact. On the other hand, gases show more adhesive than cohesive properties. This is why gases stick easily to other surfaces.
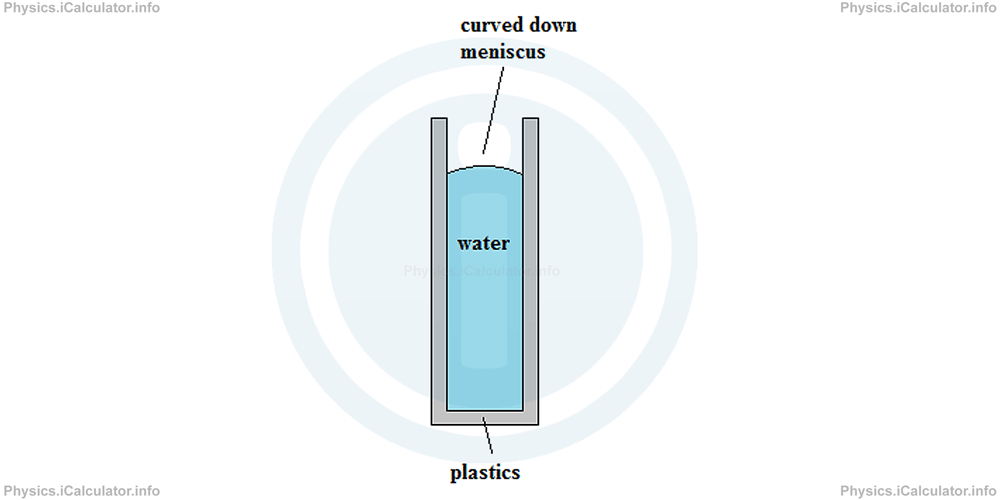
Liquids, as an intermediate category, manifest different behaviour depending on the surfaces they are in contact with. Thus, water (as the most widespread liquid) manifests cohesive behaviour when it is in contact with less dense materials such as plastics (most plastics have a smaller density than water, i.e. less than 1000 kg/m3). For example, when we pour some water inside a narrow tube made of plastics, water molecules stick to themselves more than to the plastics surface. As a result, the water surface curves down as shown in the figure.
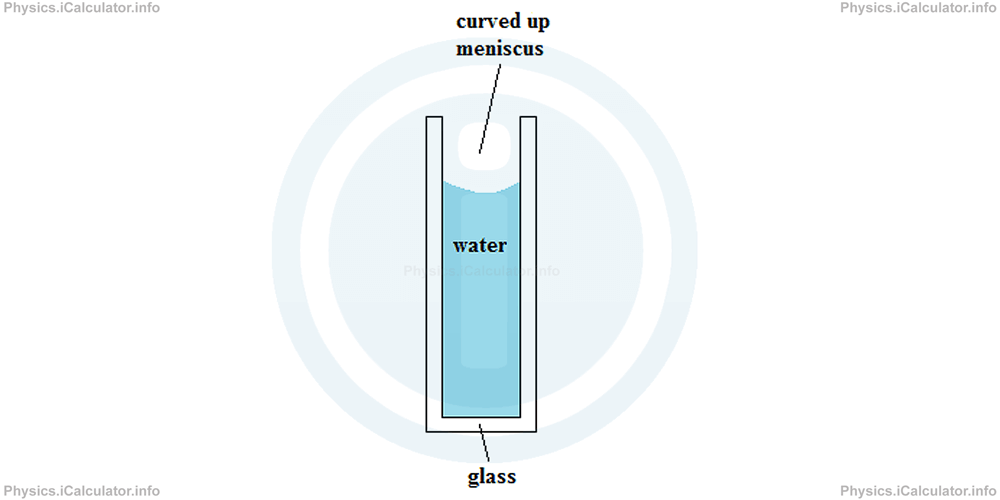
On the other hand, when liquids are in contact with a denser substance, they manifest more adhesive than cohesive properties. As an example, we can mention water when poured in a glass tube (glass has a density of 2500 kg/m3). In such cases, the meniscus curves up, as water tends to stick on container walls.
When adhesive and cohesive forces are equal, the liquid surface is entirely horizontal.
You have reached the end of Physics lesson 9.7.1 Adhesive and Cohesive Forces. There are 3 lessons in this physics tutorial covering Adhesive and Cohesive Forces. Surface Tension and Capillarity, you can access all the lessons from this tutorial below.
More Adhesive and Cohesive Forces. Surface Tension and Capillarity Lessons and Learning Resources
Whats next?
Enjoy the "Adhesive and Cohesive Forces" physics lesson? People who liked the "Adhesive and Cohesive Forces. Surface Tension and Capillarity lesson found the following resources useful:
- Definition Feedback. Helps other - Leave a rating for this definition (see below)
- Density and Pressure Physics tutorial: Adhesive and Cohesive Forces. Surface Tension and Capillarity. Read the Adhesive and Cohesive Forces. Surface Tension and Capillarity physics tutorial and build your physics knowledge of Density and Pressure
- Density and Pressure Revision Notes: Adhesive and Cohesive Forces. Surface Tension and Capillarity. Print the notes so you can revise the key points covered in the physics tutorial for Adhesive and Cohesive Forces. Surface Tension and Capillarity
- Density and Pressure Practice Questions: Adhesive and Cohesive Forces. Surface Tension and Capillarity. Test and improve your knowledge of Adhesive and Cohesive Forces. Surface Tension and Capillarity with example questins and answers
- Check your calculations for Density and Pressure questions with our excellent Density and Pressure calculators which contain full equations and calculations clearly displayed line by line. See the Density and Pressure Calculators by iCalculator™ below.
- Continuing learning density and pressure - read our next physics tutorial: Fluids. Density of Fluids
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "Adhesive and Cohesive Forces. Surface Tension and Capillarity" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.