Menu
Physics Lesson 5.1.5 - How is Energy classified?
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
Welcome to our Physics lesson on How is Energy classified?, this is the fifth lesson of our suite of physics lessons covering the topic of Work and Energy. Types of Energy, you can find links to the other lessons within this tutorial and access additional physics learning resources below this lesson.
How is Energy classified?
Energy is classified in two main groups: I. Energy of state and II. Energy of transfer.
The first group (Energy of state) includes all types of energy that are possessed by the system regardless of what happens outside it. The second group (Energy of transfer), includes all types of energy exchanged between two or more systems when they enter in contact with each other.
In the following section, we will show a diagram where all types of energy are represented based on a hierarchic structure.
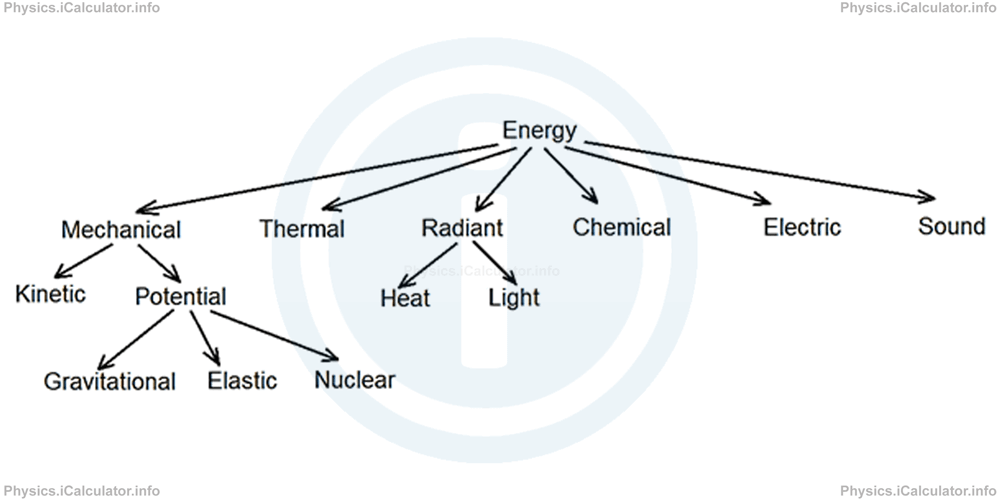
This is only a version of Energy classification; it does not represent the absolute truth. For example, Heat energy can also be included as a sub-category of Thermal energy etc.
Now, let's take a closer look at all the above mentioned types of energy. Thus,
Kinetic Energy
Kinetic Energy represents the energy a system possesses due to its motion. This means when a system is moving it possesses Kinetic Energy while when the system is at rest, it does not possess Kinetic energy. This energy can transfer from one place to another. Therefore, Kinetic Energy is an energy of transfer. You can view more detailed information on Kinetic Energy Here
Potential Energy
Potential Energy represents the energy stored in a system when no change of system's structure is considered. This stored energy can be converted into other forms of energy in certain conditions. Therefore, Potential Energy is an energy of state.
There are many kinds of Potential Energy such as:
- Gravitational Potential Energy is the stored energy in an object when it is raised at a certain height from the ground. It is obvious that when the object is released, this Gravitational Potential Energy is converted into Kinetic Energy as the object approaches the ground and loses height because it moves faster and faster.
- Elastic Potential Energy is the stored energy in an elastic object (spring, rubber band, etc.) when the object is stretched or compressed. When the elastic object is released, the Elastic Potential Energy is converted into other forms of energy as well.
- Nuclear Potential Energy is the stored energy in the nuclei of atoms. When particles from outside hit the nuclei, this stored energy is released and as a result, it turns into other forms of energy.
Mechanical Energy
Mechanical Energy represent the sum of Kinetic and Potential Energy of a system.
Thermal Energy
Thermal Energy is a part of the internal energy of a system (the other part is the chemical energy). Thermal energy is the energy possessed by the system due to the vibration of its particles (atoms and molecules). More vibrant the molecules of a system, greater the value of its thermal energy. Thermal energy is an energy of state.
Radiant Energy
Radiant Energy is the energy radiated in the environment by hot objects. Hotter the source object, greater the amount of the energy radiated around. When the radiant source is not very hot, we perceive the radiant energy as Heat and when the source is very hot, it becomes luminescent and therefore it emits radiation in the form of Light Energy. Radiant energy is an energy of transfer.
Chemical Energy
Chemical Energy is the energy released (or absorbed) by a system during a chemical reaction, i.e. when the structure of substances involved in the process changes. This is the main difference between Potential and Chemical Energy (in potential energy there is no change in the structure of material or substance). Chemical energy is an energy of state when not activated yet. Then, during a chemical reaction it is transferred.
Electric Energy
Electric Energy is the energy produced when the electrons of a conductor flow in a pre-defined direction. It is an Energy of transfer.
Sound Energy
Sound energy is produced when a sound source disturbs the medium's molecules (usually air molecules) and make them vibrate. This disturbance is propagated in the medium in the form of sound waves until it reaches the receiver. Therefore, it is obvious that sound energy is a kind of transfer energy.
You have reached the end of Physics lesson 5.1.5 How is Energy classified?. There are 5 lessons in this physics tutorial covering Work and Energy. Types of Energy, you can access all the lessons from this tutorial below.
More Work and Energy. Types of Energy Lessons and Learning Resources
Whats next?
Enjoy the "How is Energy classified?" physics lesson? People who liked the "Work and Energy. Types of Energy lesson found the following resources useful:
- Classification Feedback. Helps other - Leave a rating for this classification (see below)
- Work, Energy and Power Physics tutorial: Work and Energy. Types of Energy. Read the Work and Energy. Types of Energy physics tutorial and build your physics knowledge of Work, Energy and Power
- Work, Energy and Power Revision Notes: Work and Energy. Types of Energy. Print the notes so you can revise the key points covered in the physics tutorial for Work and Energy. Types of Energy
- Work, Energy and Power Practice Questions: Work and Energy. Types of Energy. Test and improve your knowledge of Work and Energy. Types of Energy with example questins and answers
- Check your calculations for Work, Energy and Power questions with our excellent Work, Energy and Power calculators which contain full equations and calculations clearly displayed line by line. See the Work, Energy and Power Calculators by iCalculator™ below.
- Continuing learning work, energy and power - read our next physics tutorial: Kinetic Energy
Help others Learning Physics just like you
Please provide a rating, it takes seconds and helps us to keep this resource free for all to use
We hope you found this Physics lesson "Work and Energy. Types of Energy" useful. If you did it would be great if you could spare the time to rate this physics lesson (simply click on the number of stars that match your assessment of this physics learning aide) and/or share on social media, this helps us identify popular tutorials and calculators and expand our free learning resources to support our users around the world have free access to expand their knowledge of physics and other disciplines.